Colorectal Cancer: Identifying your at-risk participants and the business impact
For employers, health plans and providers, keeping your participants healthy is one of your highest priorities. This especially rings true with a preventable disease like colorectal cancer.
In fact, the vast majority of colorectal cancers could be successfully treated if everyone eligible were screened. By knowing which participants are at risk and recommending regular screenings to those participants, it is possible to improve both early detection rates and survivability. Epidemiologic data from the Surveillance, Epidemiology and End Results Program (SEER) suggests that the 5-year survival rate for localized colorectal cancer is above 90%.
Let’s discuss the types of screening options available, shed light on who, among your participants, should be prioritized for colorectal cancer screenings and pinpoint the business impact of colorectal cancer on your organization.
Evaluating testing options
From invasive to non-invasive, to procedures conducted in a hospital setting or samples collected at home — there are many screening options to help your participants detect colon cancer early, including the Everlywell at-home colon test (fecal immunochemical test).
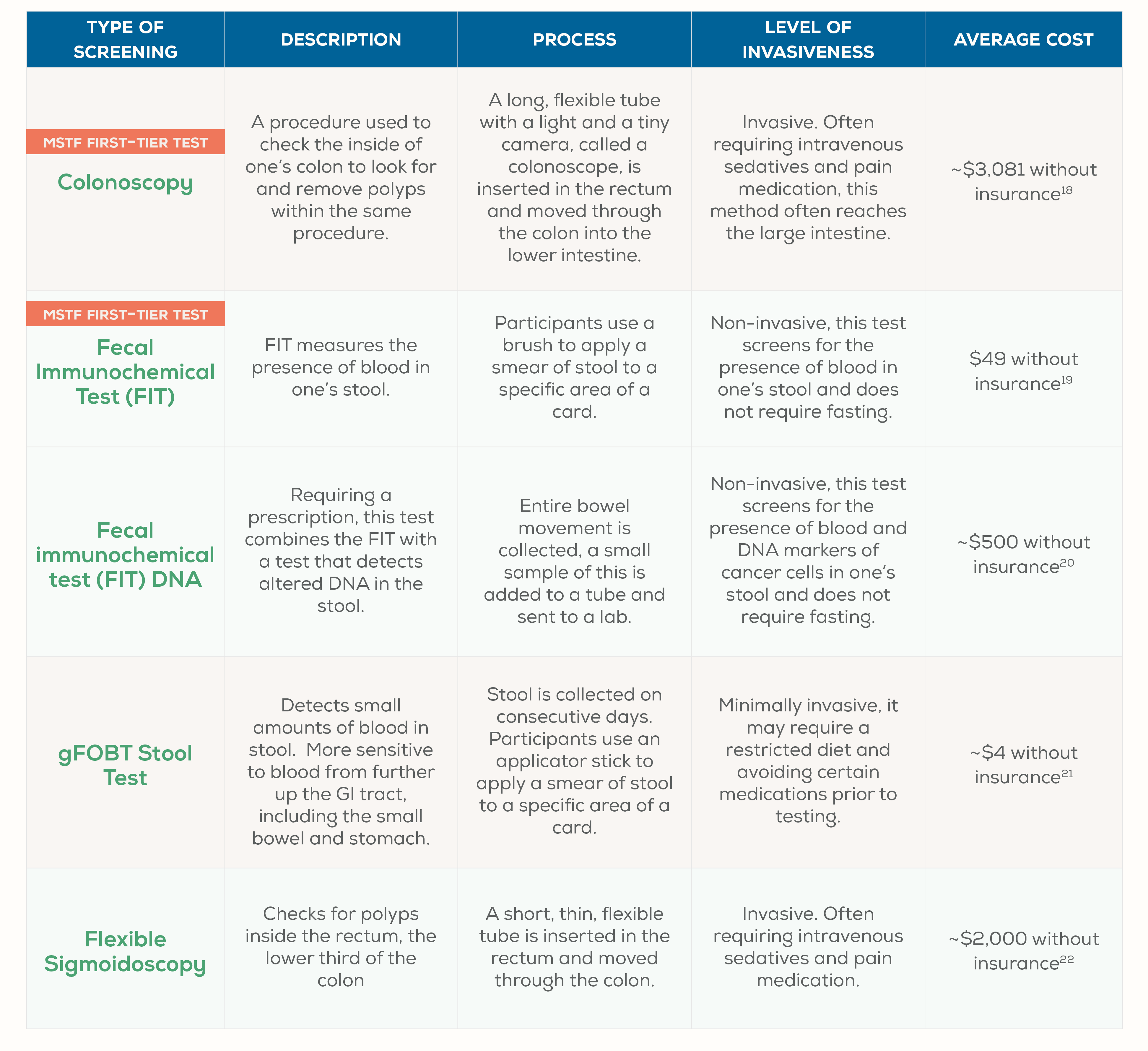
Risk segments
Now that we have a good idea of the types of tests available, let’s examine the three most important risk stratifications that your participants may represent. Note that these are population-based guidances.
Segment 1: Participants age 45-75
USPSTF recommended options for colorectal cancer screening

Segment 2: Participants age 76 and older
| Age | Recommendation |
|---|---|
| Up to age 85 | Individuals who have never received prior screening for colorectal cancer should be screened (with consideration for age, comorbidities, and life expectancy). |

Segment 3: High risk participants

Examples of high risk individuals:
Patients with:
- Lynch Syndrome or familial adenomatous polyposis
- Inflammatory bowel disease (ulcerative colitis or Crohn's disease)
- Personal or family history of colon cancer, adenomas, or other related cancers (multiple family members increase risk)
- A positive result from another screening method in the last year
Type of test recommended and at what frequency
Individuals who meet any of the criteria previously mentioned and are considered high risk for developing colorectal cancer should consult a gastroenterologist about when to initiate colorectal cancer screening, what type of screening test is most appropriate for them, and how frequently they should repeat screening. The age of initial screening and surveillance intervals are highly dependent on the nature of the risk. The U.S. Multi-Society Task Force for Colorectal Cancer documents many of these risk-modifying attributes and the adjusted screening recommendations. For instance, if an individual has a single first-degree relative diagnosed with colorectal cancer at age >60, they should have their initial screening at age 40.
Why this segment is important
Risk is assessed in many ways - both statistically and qualitatively. These recommendations are based on epidemiologic research, population based studies, and consensus of experts in the field of colorectal cancer prevention. Within this context, the evidence suggests that high-risk individuals require earlier and/or more frequent screening.
For employers and health plans: Cost and patient experience
Calculating the cost of colorectal cancer
According to the CDC, the total annual medical cost of colorectal cancer care in the U.S. is $14.1 billion[10]. This represents the second-highest cost of any cancer in the United States. On average, Medicare healthcare spending for patients with newly diagnosed colorectal cancer ranges from $40,000-80,000 depending on the stage of the cancer[11]. With the Kaiser Family Foundation reporting that roughly 60% of U.S. companies partially or fully self-fund their health plans[12], an employee with colon cancer can substantially raise insurance premiums and eliminate self-insurance reserves. In fact, a study of major employers found that participants with cancer cost more than 5x as much to insure than those without cancer (around $16,000 vs. $3,000 each annually)[13].
There’s also the impact on employee productivity. A National Cancer Institute-funded study simulated a reduction in colorectal cancer risk factors and increasing screening and treatment from 2005 to 2020. The study found that 101,353 deaths could be avoided and $33.9 billion saved in reduced productivity loss. The same study modeled that increased screening rates alone could result in almost $14.7 billion in savings between 2005 and 2020[14].
The bottom line? With regular screening programs for colorectal cancer, employers can play a pivotal role in saving lives and reducing health care costs. Learn more about how Everlywell is helping employers screen their employees (including the at-home colon cancer test) with at-home lab tests here.

Improving patient experience measures
Colorectal Cancer Screening is a required Healthcare Effectiveness Data and Information Set (HEDIS) measure[15]. It assesses whether adults age 50–75 undergo the above-mentioned screenings at their recommended cadence. By engaging your members with colorectal cancer screening options such as an annual FIT test, you are improving the overall quality of care provided to your members. An at-home lab test with a high Net Promoter Score (NPS) like the Everlywell FIT colorectal cancer screening test can also help create a positive member experience. As patient experience and satisfaction become increasingly adopted performance metrics for plans and providers, it is important to engage in programs that directly benefit these measures. In addition, at-home lab tests are a convenient and effective solution for closing gaps in care, helping your plan increase compliance rates.
In a 2011 study of over 3.2 million members, Kaiser Permanente began using targeted mail outreach to encourage their participants to participate in FIT screening. The results of this outreach, combined with opportunistic screening measures, resulted in the proportion of the commercially insured population screened in accordance with HEDIS measures at Kaiser increased from 37 to 69% from 2005 to 2010, and the proportion of the Medicare population screened increased from 41 to 78%. Unpublished data indicate this increase in screening has been associated with a change in cancer stage and even the incidence of colorectal cancer among the Kaiser population.
By focusing on the patient experience and encouraging participation in opportunistic screening, you will significantly increase screening rates, benefit member experience, and improve health outcomes.
To learn more about the risks of colorectal cancer among your participants, check out our white paper.
References
Bankrate. How much does a colonoscopy cost?. Date accessed: 3/1/21
Everlywell. Preventative colon cancer screening from home. Date accessed: 3/1/21.
Gastroenterology Consultants of San Antonio. The Truth About Cologuard Tests. Date accessed: 3/9/21
Medimpex. FOBT - Home Fecal Occult Blood Test (Qty 1). Date accessed: 3/25/21.
MD Save. Sigmoidoscopy / Proctosigmoidoscopy. Date accessed: 3/9/21.
NCBI. Colorectal Cancer Screening: Recommendations for Physicians and Patients from the U.S. Multi-Society Task Force on Colorectal Cancer. Date accessed: 3/5/21. do not differentiate among
SEER Cancer Stat Facts: Colorectal Cancer. National Cancer Institute. Date Accessed: 2/28/21
U.S. Bureau of Labor Statistics. Older workers: Labor force trends and career options. 5/17.
The American Journal of Gastroenterology. Colorectal Cancer Screening: Recommendations for Physicians and Patients from the U.S. Multi-Society Task Force on Colorectal Cancer. July 2017.
CDC. Cost-Effectiveness of Colorectal Cancer Interventions. 9/29/20.
CDC. Cost-Effectiveness of Colorectal Cancer Interventions. 9/29/20.
Kaiser Family Foundation. 2017 Employer Health Benefits Survey. Accessed 3/9/21
CEO Roundtable on Cancer. Making the Business Case: How Engaging Employees in Preventive Care Can Reduce Healthcare Costs. Date Accessed: 3/9/21.
NCBI. Productivity Savings from Colorectal Cancer Prevention and Control Strategies. Date Accessed: 3/9/21.
NCQA. Required HEDIS® and CAHPS® Measures for HEDIS Reporting Year 2021. Date Accessed: 3/9/21